 Thus, when an electron is in its ground state, it has lower energy than when it is in its excited state. When the electron is excited to the next energy level, it goes to an orbital with higher energy. This is because, in the ground state, the electron is in the lowest energy orbital (i.e. The energy level diagram of the hydrogen atom clearly shows that the ground state energy level (E0) is lower than the first excited state energy level (E). The energy levels are shown as horizontal lines and the orbitals are shown as arrows. The energy level diagram of a hydrogen atom is as follows: The simplest example for an energy level diagram is that of a hydrogen atom that has only one electron. Energy level diagrams can be drawn for molecules as well as for atoms. In other words, energy level diagrams depict the energies of molecular orbitals (MOs) in a molecule. Representation of energy level diagrams:Įnergy level diagrams are the pictorial representations of energy levels of electrons in an atom or molecule. The possible values an electron can have for any given energy level are also quantized. The energies of photons emitted by an atom are always quantized, which means they can only have certain values. 
The energy of a photon is equal to the difference in energy between the two energy levels involved in the transition. 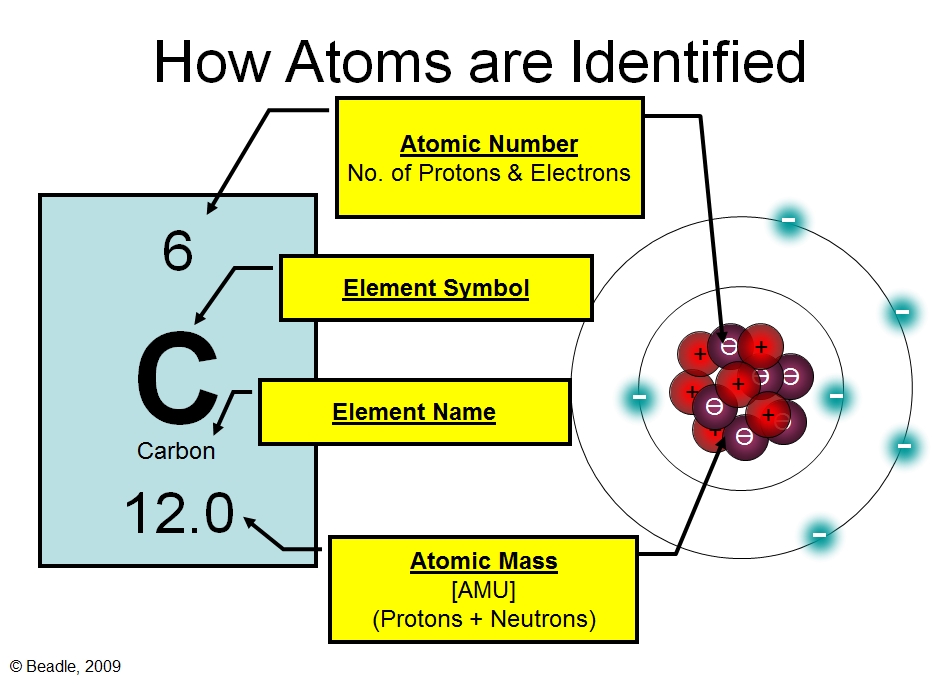
The most important feature of an energy level diagram is the line between two energies called an “energy transition.”Įnergy transitions correspond to photons being emitted or absorbed by an atom. In an energy level diagram, the horizontal axis represents the energy of an electron and the vertical axis represents the number of electrons present at that energy. To understand the light emitted by atoms, we need to understand energy level diagrams. The horizontal axis represents the orbital energy, and the vertical axis represents the total energy of the system. The blue line represents the ground state energy, and the red line represents the excited state energy. 
The molecular orbitals are shown as horizontal lines, and the energies are shown as vertical lines. This type of diagram shows the energies of the orbitals in a molecule. The most common type of energy level diagram is the molecular orbital energy level diagram. Stay tuned for more! What is an energy level diagram?Īn energy level diagram, or energy band diagram, is a graphical representation of the allowed energy states of a system. In this blog post, we will take a closer look at the energy level diagram of the hydrogen atom and explore how it helps us understand atomic structure and bonding. The hydrogen atom is a good example to start with in order to gain a fundamental understanding of energy level diagrams. It's thought that another one of these magic numbers lies just beyond the range of currently known elements, which means we could potentially discover some new elements that are relatively stable and extremely massive.A clear depiction of the energy level diagram is important to understand the various states and transitions of energy in an atom or molecule. 
There are believed to be certain numbers/ratios that are inherently more stable. Hence, larger atoms become more unstable. The ratio of neutrons to protons gradually increases to try to compensate, but this results in the neutrons getting more unstable themselves, resulting in more opportunity for weak forces to destabilize them. With all of that to consider, as you get larger and larger nuclei with more protons, the electromagnetic forces increase, as well as the distances in the nuclei, putting more strain on the nuclear forces holding it all together. Neutrons will help add to this nuclear force, but they tend to become unstable when they get into too great of concentration and the weak force will cause them to decay into protons. Fusion allows these protons to get close enough together for the stronger, yet short ranged, nuclear force to overpower the electromagnetic force. The electromagnetic force of the protons wants to push them away from each other. There are several forces at work inside the nuclei of atoms. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
